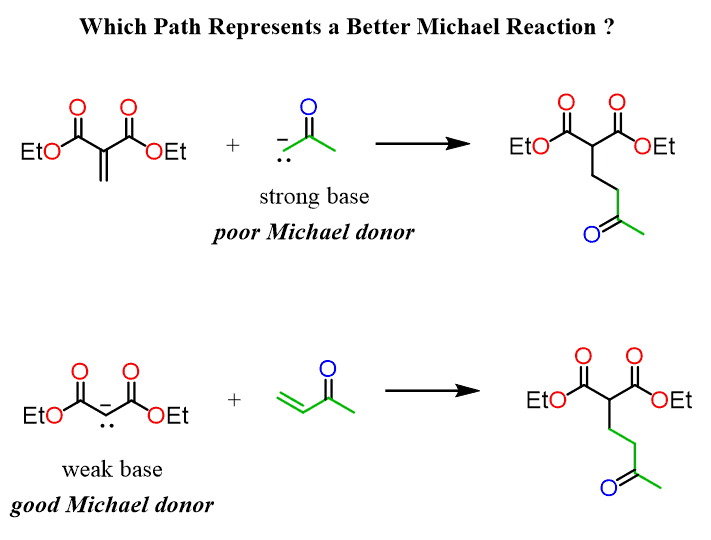
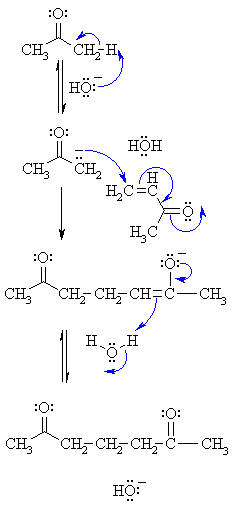
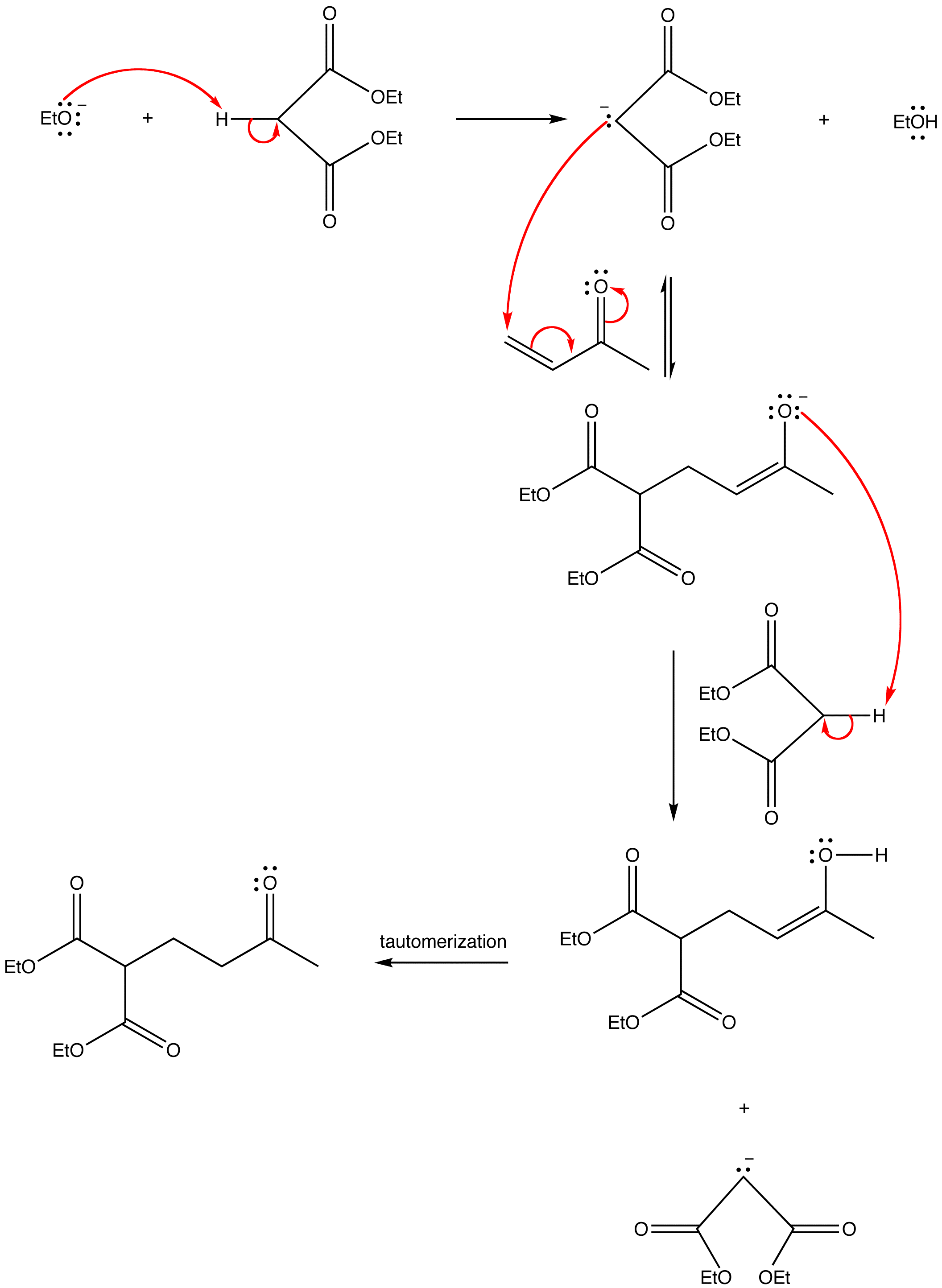
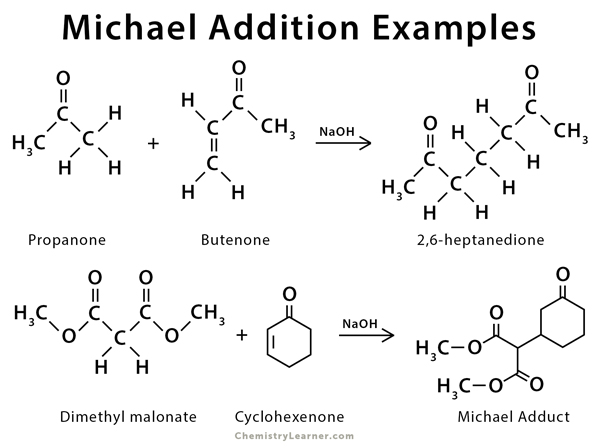
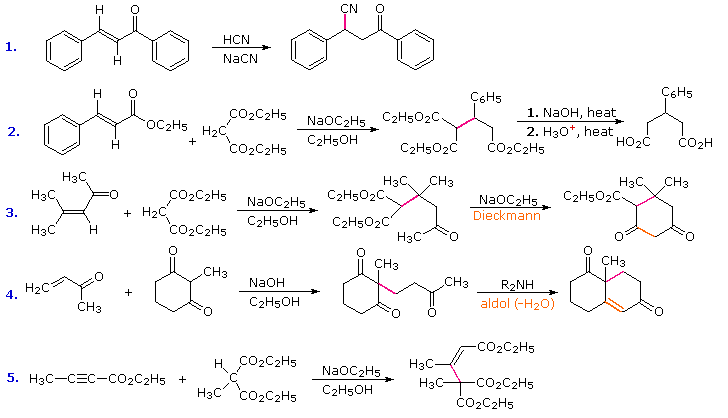
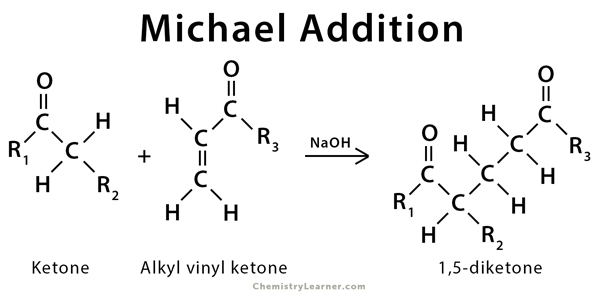
Solvent-free, catalyst-free aza-Michael addition of cyclohexylamine to diethyl maleate: Reaction mechanism and kinetics - ScienceDirect

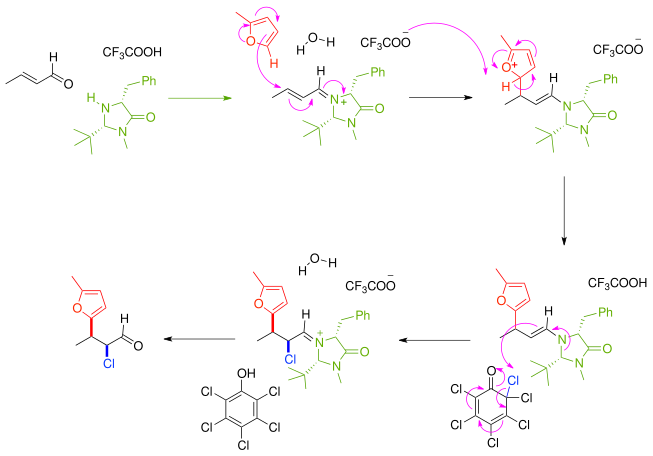
Enantioselective Michael Addition of Aldehydes to β‐Nitrostyrenes Catalyzed by (S)‐N‐(D‐Prolyl)‐1‐triflicamido‐3‐phenylpropan‐2‐amine - Gorde - 2019 - European Journal of Organic Chemistry - Wiley Online Library