
Thioflavin T as an amyloid dye: fibril quantification, optimal concentration and effect on aggregation | Royal Society Open Science

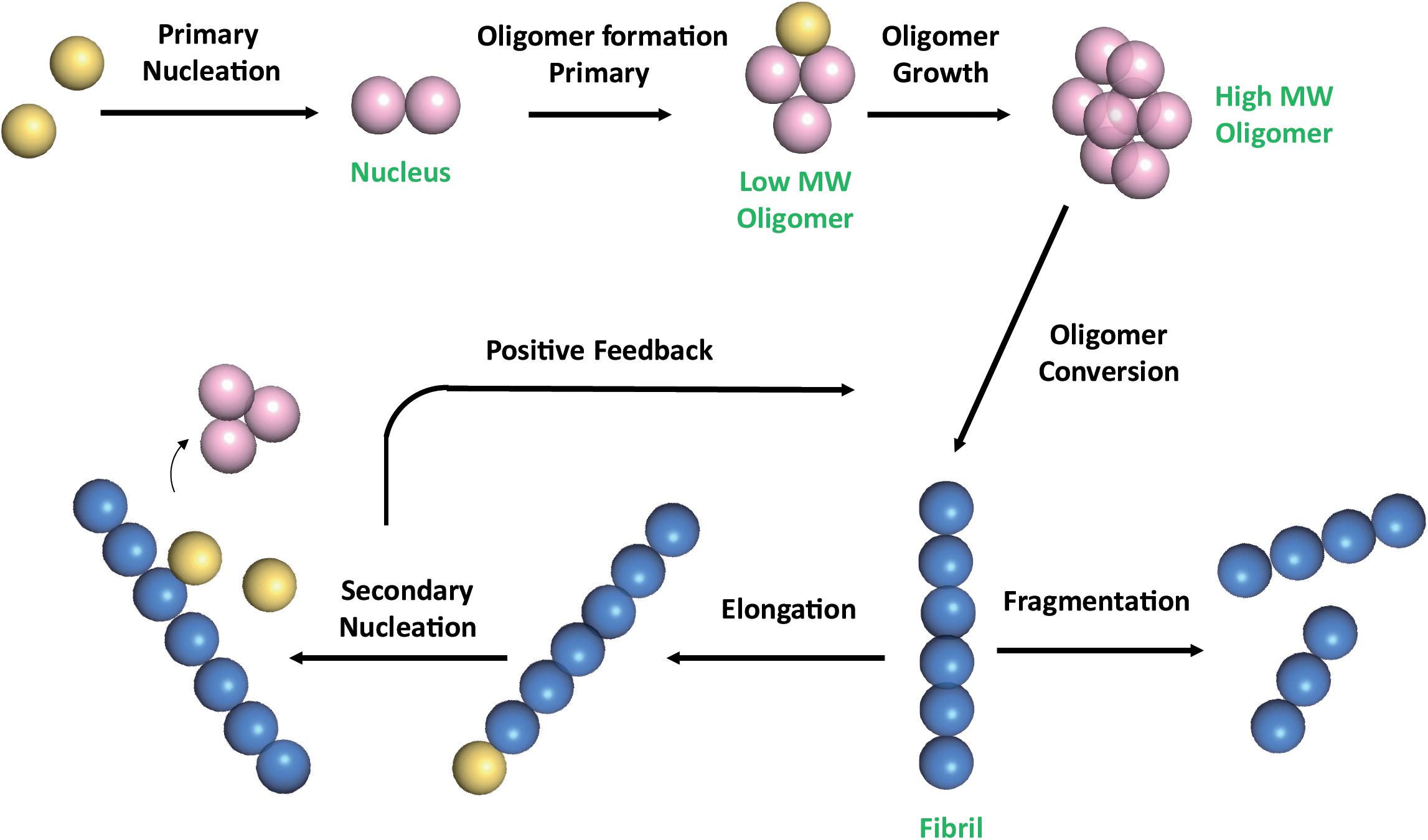
A) Schematic illustration of the nucleated pathway of amyloid fibril... | Download Scientific Diagram

Molecular Mechanism of Thioflavin-T Binding to the Surface of β-Rich Peptide Self-Assemblies - ScienceDirect
Frontiers | Natural Compounds as Inhibitors of Aβ Peptide Aggregation: Chemical Requirements and Molecular Mechanisms | Neuroscience

Kinetic Mechanism of Thioflavin T Binding onto the Amyloid Fibril of Hen Egg White Lysozyme | Langmuir
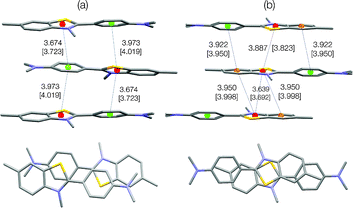
Crystal structure of thioflavin-T and its binding to amyloid fibrils : insights at the molecular level - Chemical Communications (RSC Publishing) DOI:10.1039/B912396B
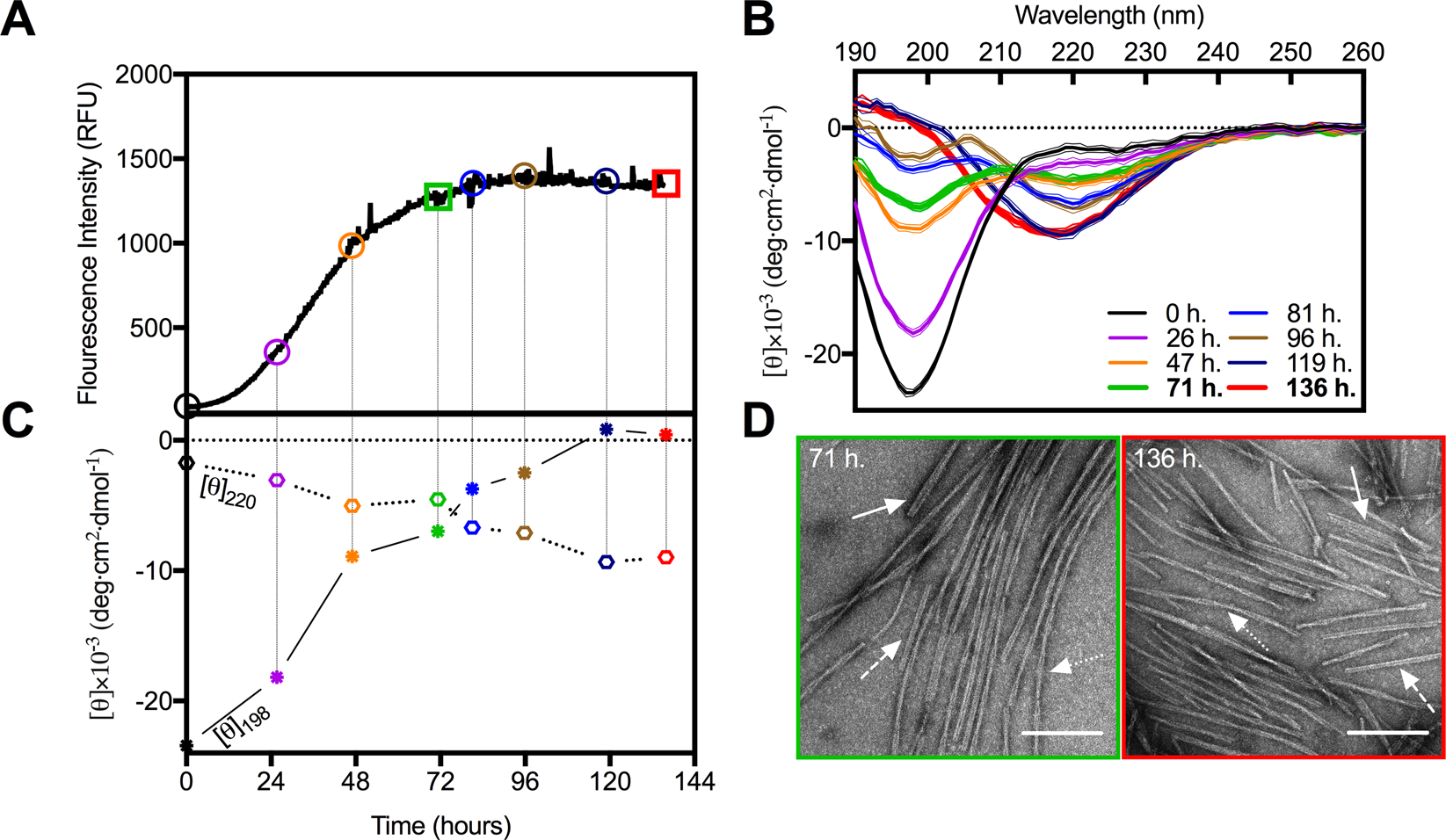
Early Stage Alpha-Synuclein Amyloid Fibrils are Reservoirs of Membrane- Binding Species | Scientific Reports
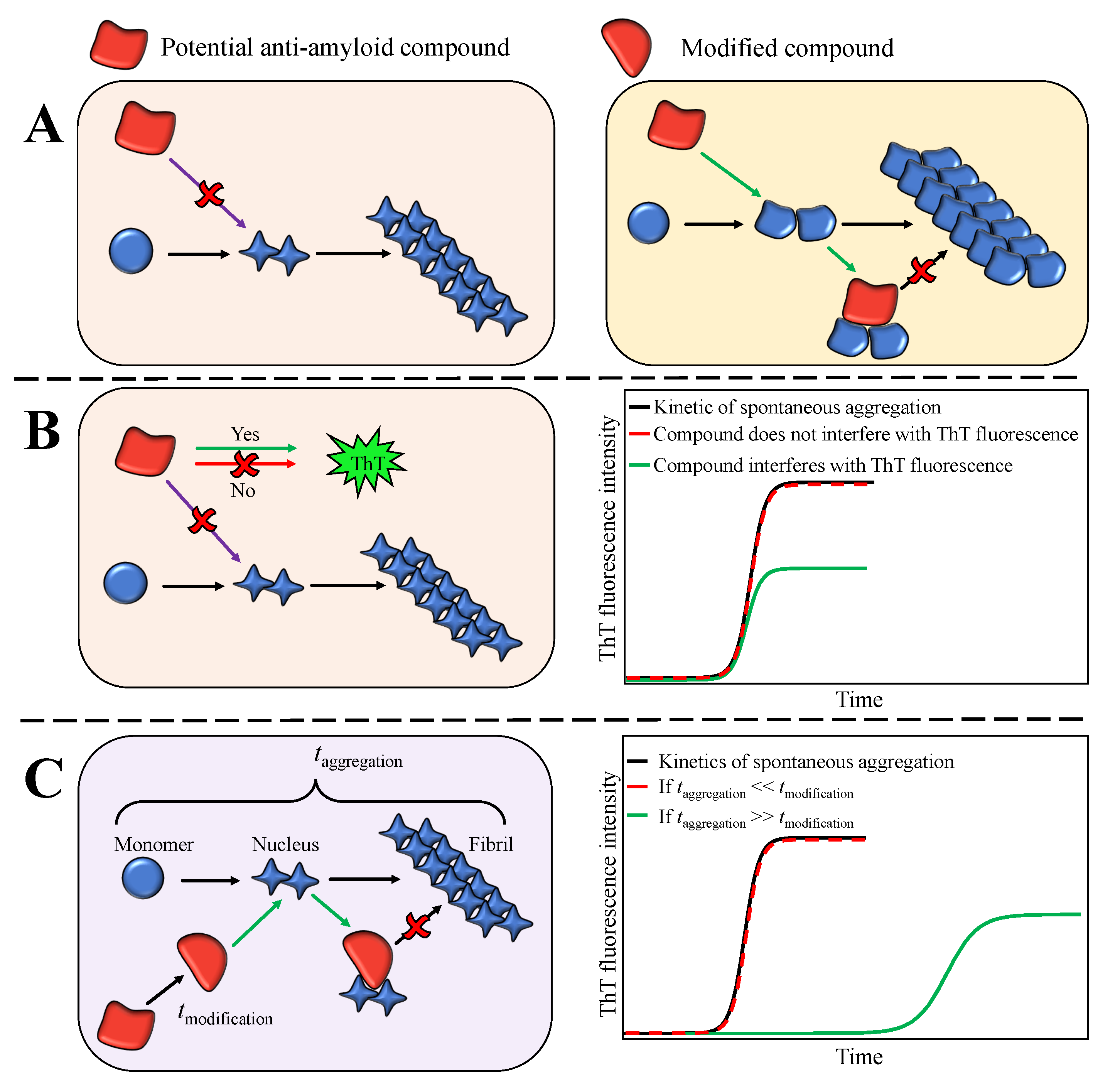
Biomolecules | Free Full-Text | The Environment Is a Key Factor in Determining the Anti-Amyloid Efficacy of EGCG | HTML

Modulation of the extent of structural heterogeneity in α-synuclein fibrils by the small molecule thioflavin T - Journal of Biological Chemistry
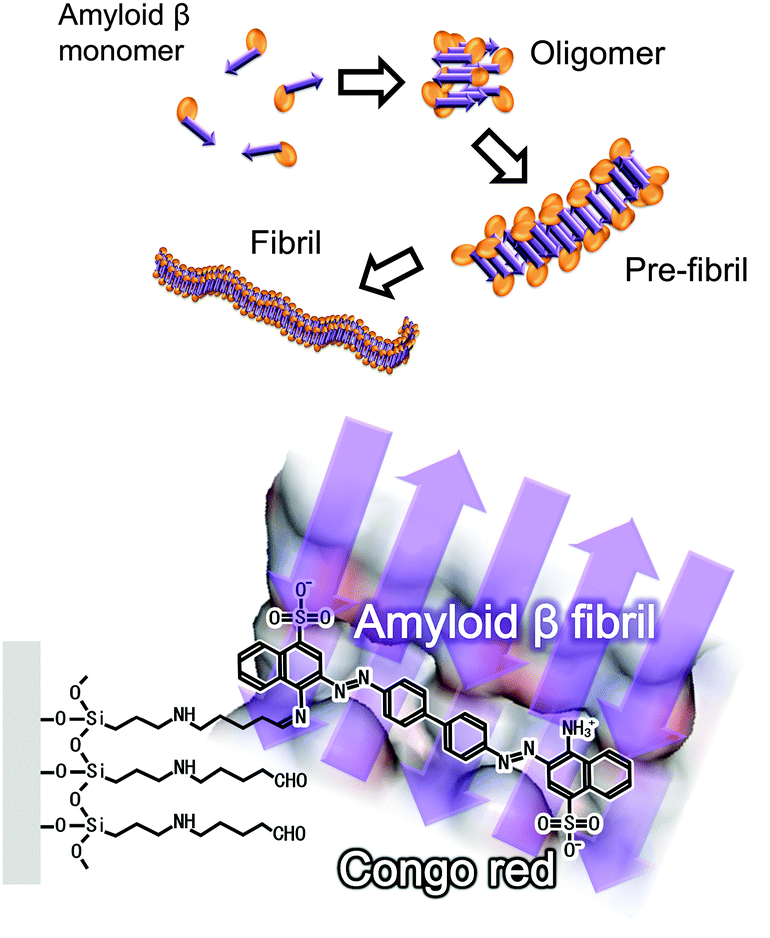
A label-free electrical assay of fibrous amyloid β based on semiconductor biosensing - Chemical Communications (RSC Publishing) DOI:10.1039/C3CC49460H

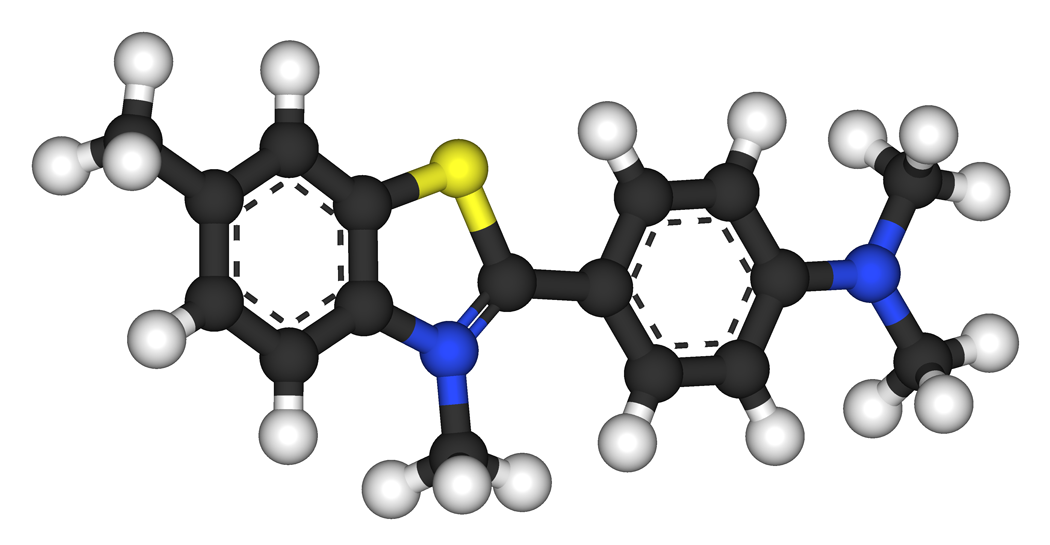







![Exploring the occurrence of thioflavin-T-positive insulin amyloid aggregation intermediates [PeerJ] Exploring the occurrence of thioflavin-T-positive insulin amyloid aggregation intermediates [PeerJ]](https://dfzljdn9uc3pi.cloudfront.net/2021/10918/1/fig-1-full.png)

![Fibrillation mechanism controlled by CB[6]. (a) Thioflavin T (ThT)... | Download Scientific Diagram Fibrillation mechanism controlled by CB[6]. (a) Thioflavin T (ThT)... | Download Scientific Diagram](https://www.researchgate.net/publication/318504796/figure/fig3/AS:518719889985536@1500683867675/Fibrillation-mechanism-controlled-by-CB6-a-Thioflavin-T-ThT-assay-for-the-kinetics.png)