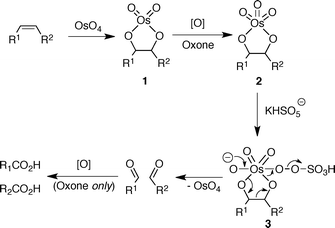
Catalytic oxidative cleavage of olefins promoted by osmium tetroxide and hydrogen peroxide - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C0OB01189D

Scheme of reactions of osmium tetroxide with unsaturated lipids and... | Download Scientific Diagram
![SOLVED:Question B4 [9 minutes] (a) Ozonolysis of styrene yields benzaldehyde and formaldehyde. Provide a mechanism for this transformation_ MezS (b) Oxidation of styrene with osmium tetroxide forms vicinal diol. Draw the structure SOLVED:Question B4 [9 minutes] (a) Ozonolysis of styrene yields benzaldehyde and formaldehyde. Provide a mechanism for this transformation_ MezS (b) Oxidation of styrene with osmium tetroxide forms vicinal diol. Draw the structure](https://cdn.numerade.com/ask_images/90c7bcabacd04c93849072ead28cf4cc.jpg)
SOLVED:Question B4 [9 minutes] (a) Ozonolysis of styrene yields benzaldehyde and formaldehyde. Provide a mechanism for this transformation_ MezS (b) Oxidation of styrene with osmium tetroxide forms vicinal diol. Draw the structure

OneClass: alkenes can be oxidized to cis-diols using either osmium tetroxide or cold alkaline potassi...
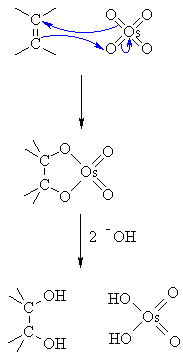
/chapter9/pages21and22/page21and22_files/diol_KMno4_mech.png)

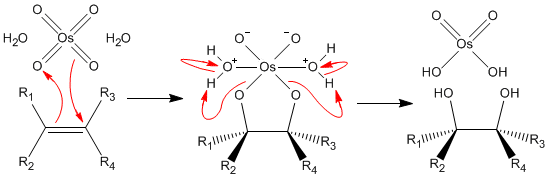


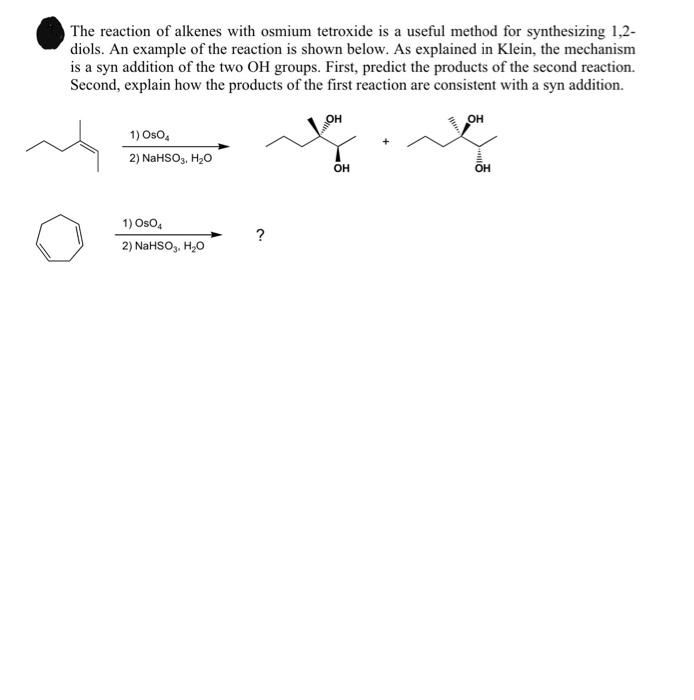
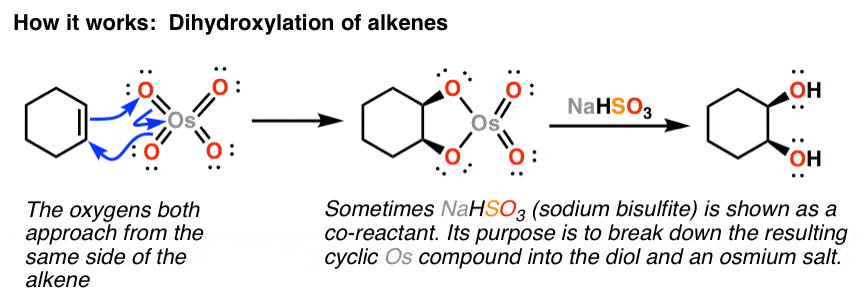



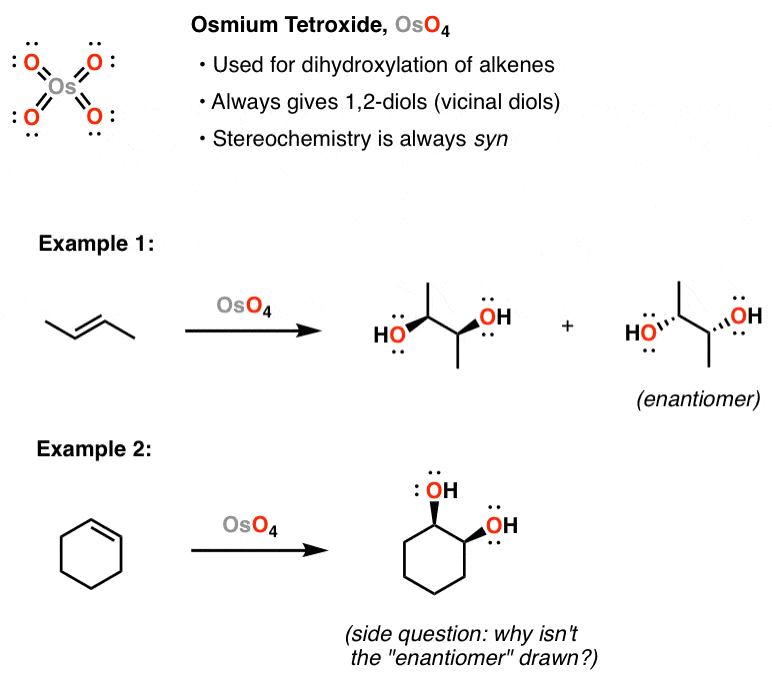
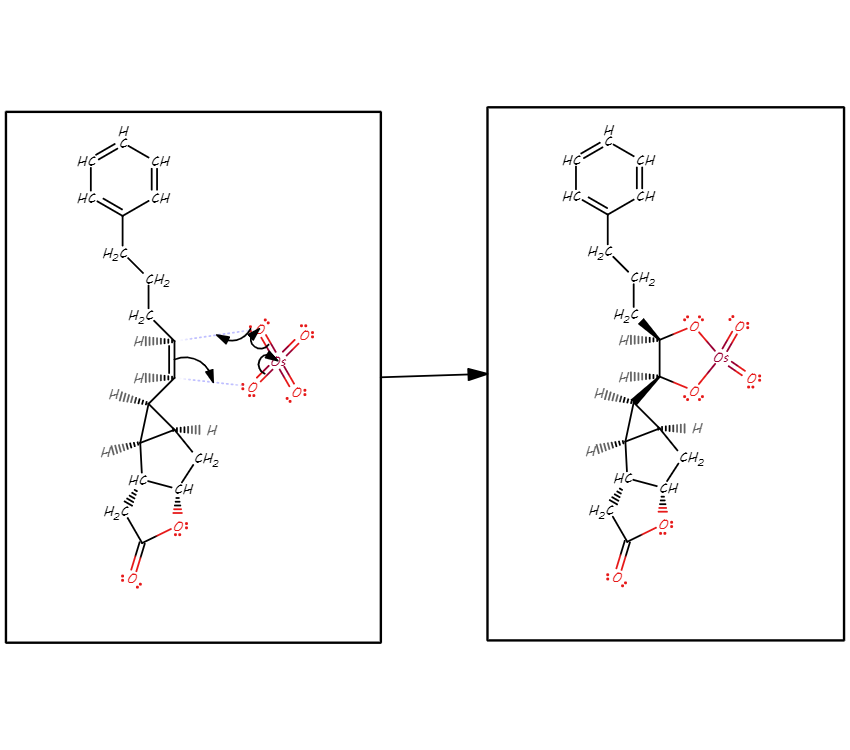

/chapter9/pages21and22/page21and22_files/diol_oso4_mech.png)
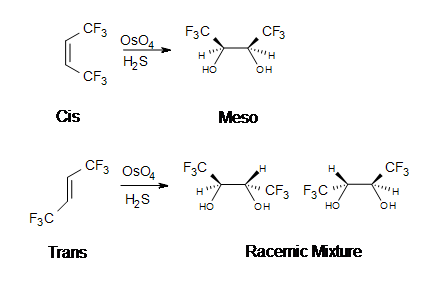


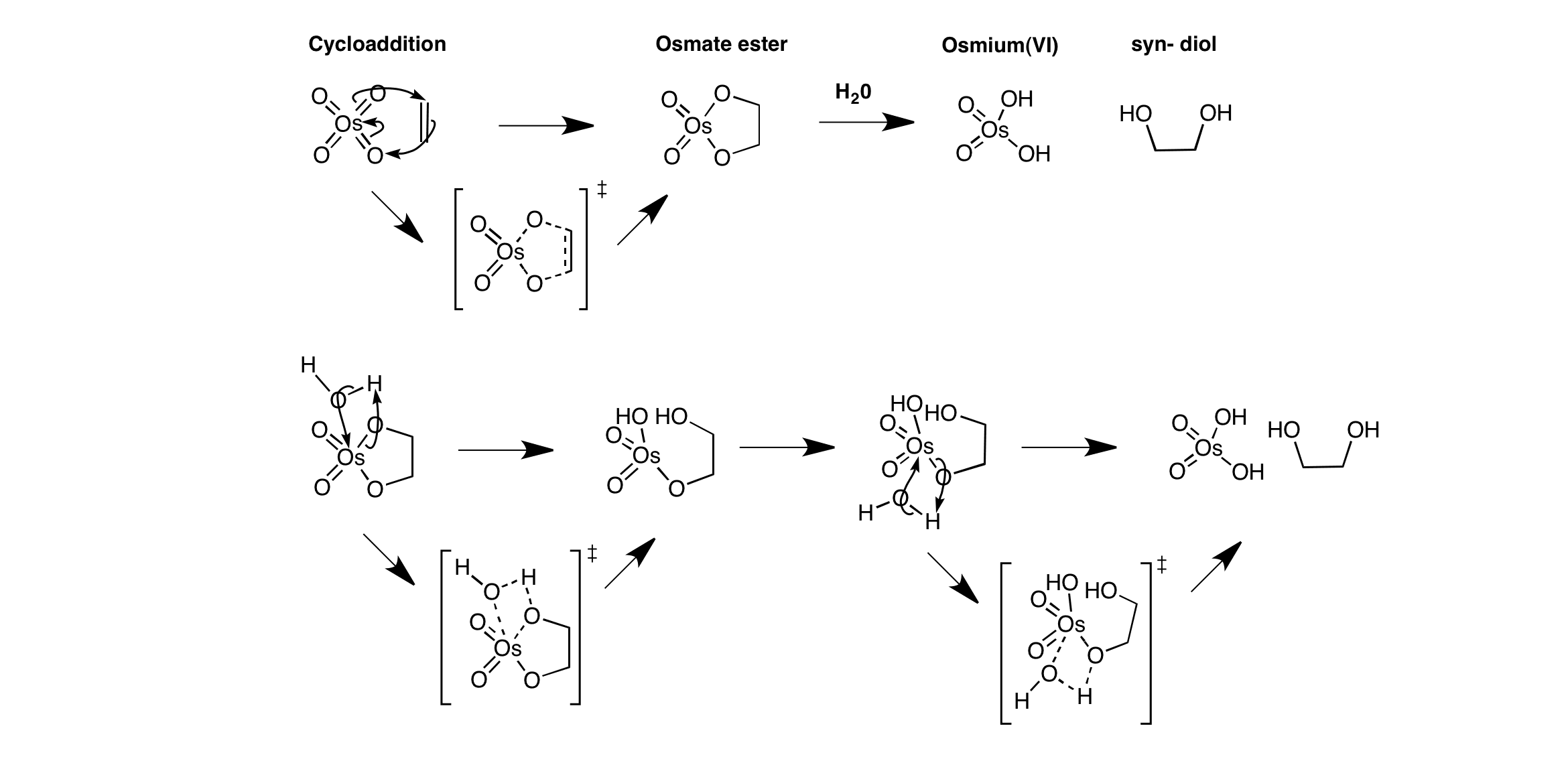
![Dihydroxylation [OsO4] - ChemistryScore Dihydroxylation [OsO4] - ChemistryScore](https://chemistryscore.com/wp-content/uploads/2019/11/Dihydroxylation1-2-768x303.png)
