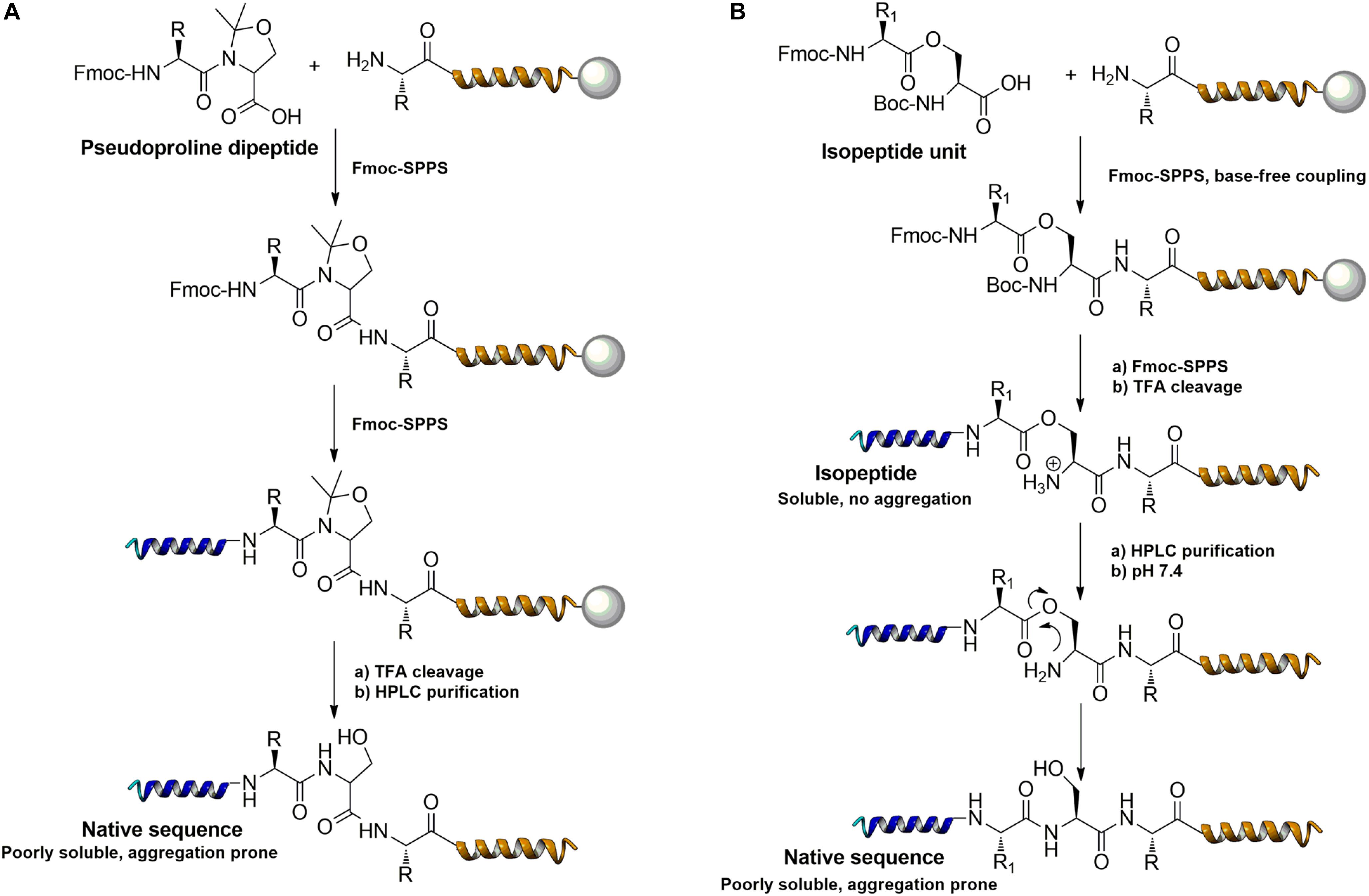
Frontiers | Challenges and Perspectives in Chemical Synthesis of Highly Hydrophobic Peptides | Bioengineering and Biotechnology
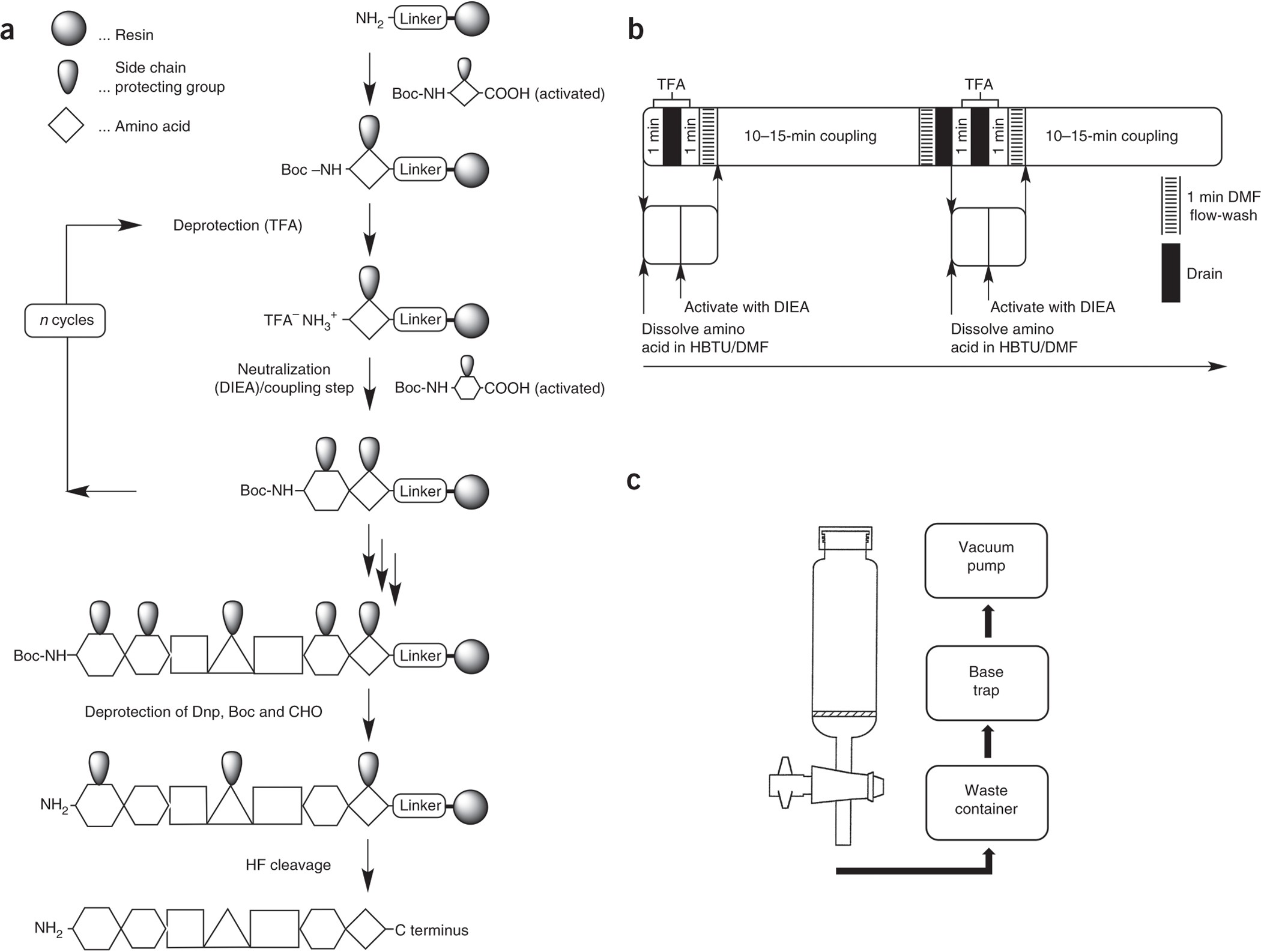
Methods, setup and safe handling for anhydrous hydrogen fluoride cleavage in Boc solid-phase peptide synthesis | Nature Protocols

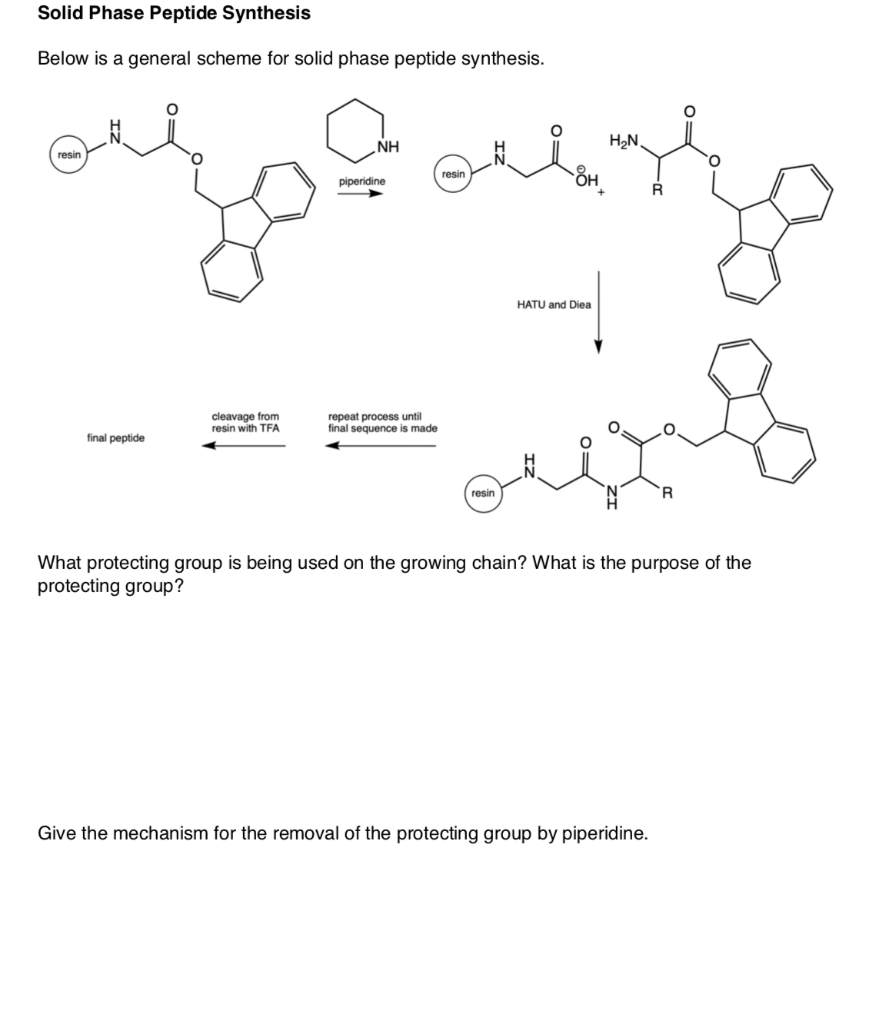
Molecules | Free Full-Text | Solid-Phase Synthetic Route to Multiple Derivatives of a Fundamental Peptide Unit | HTML

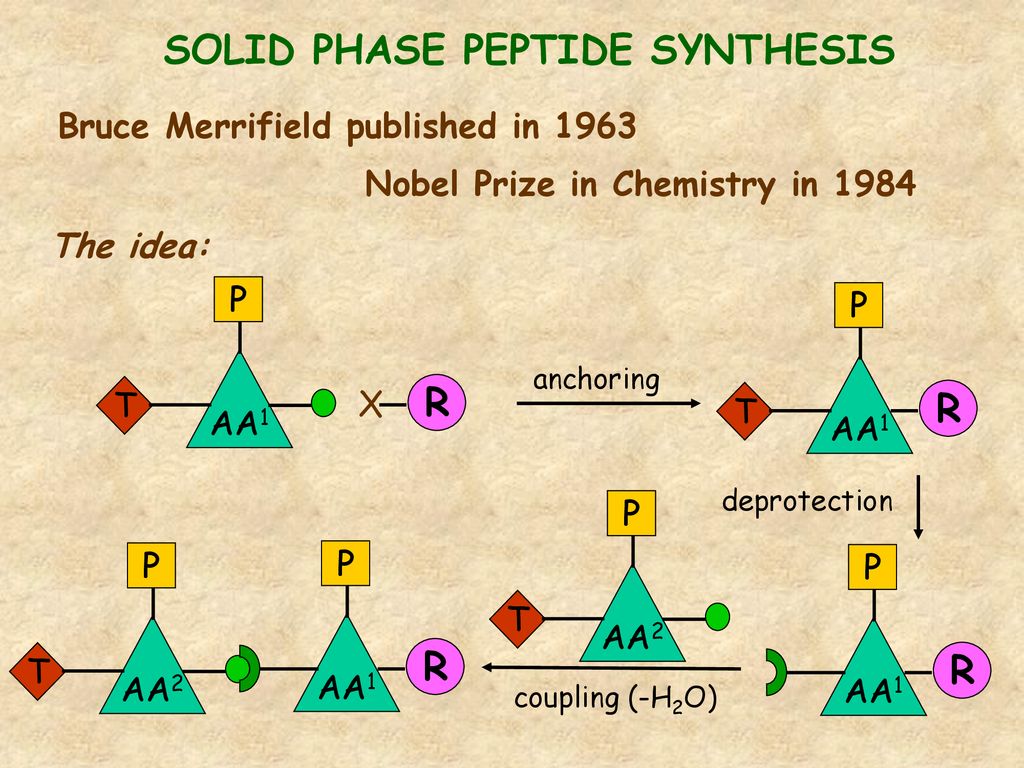
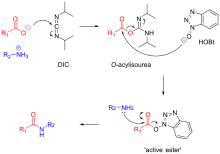
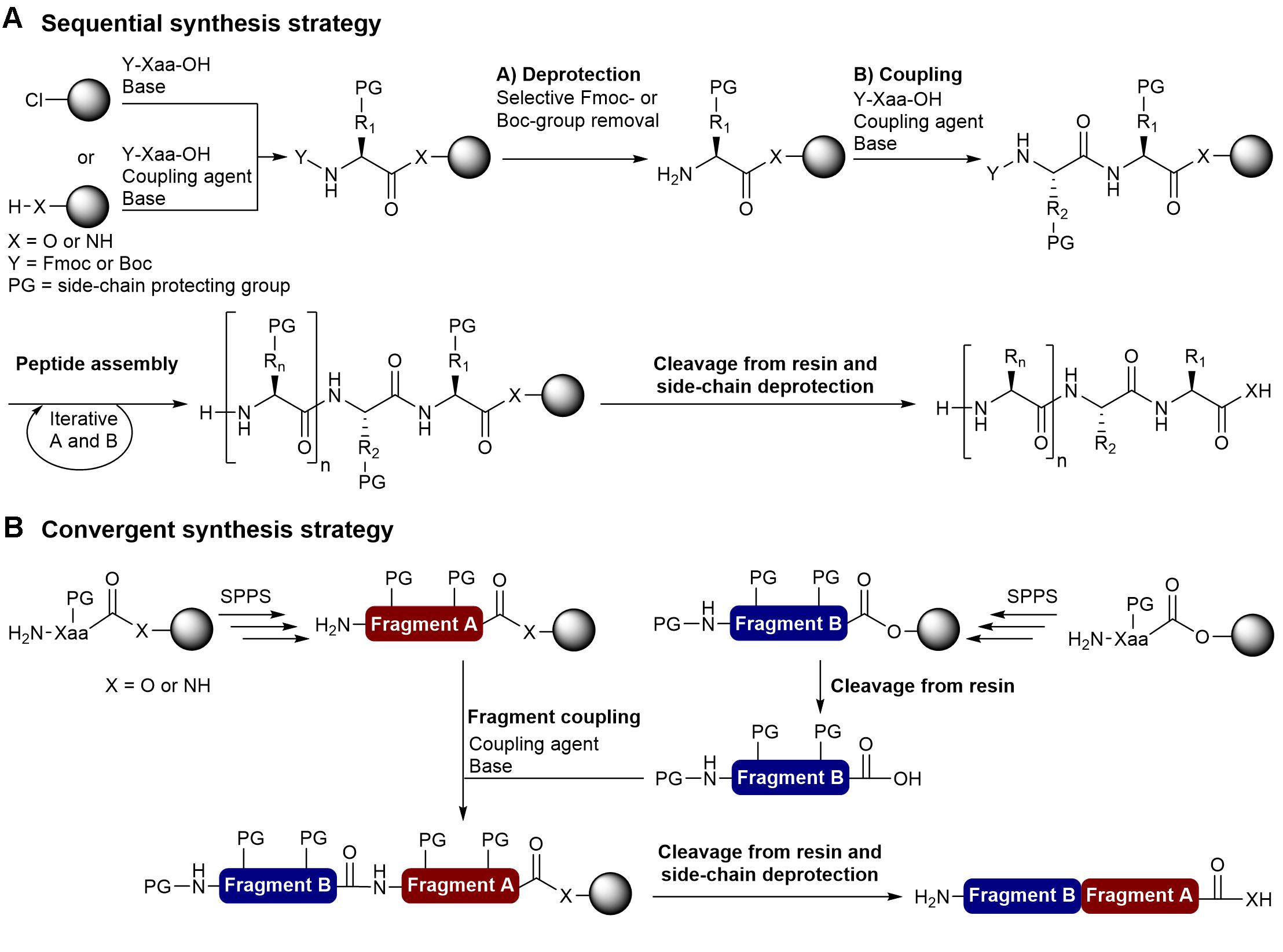
Thirteen decades of peptide synthesis: key developments in solid phase peptide synthesis and amide bond formation utilized in peptide ligation | SpringerLink

Prevention of aspartimide formation during peptide synthesis using cyanosulfurylides as carboxylic acid-protecting groups | Nature Communications
Frontiers | Recent Progress in the Chemical Synthesis of Class II and S-Glycosylated Bacteriocins | Microbiology

Positional Scanning for Peptide Secondary Structure by Systematic Solid-Phase Synthesis of Amino Lactam Peptides | Journal of the American Chemical Society
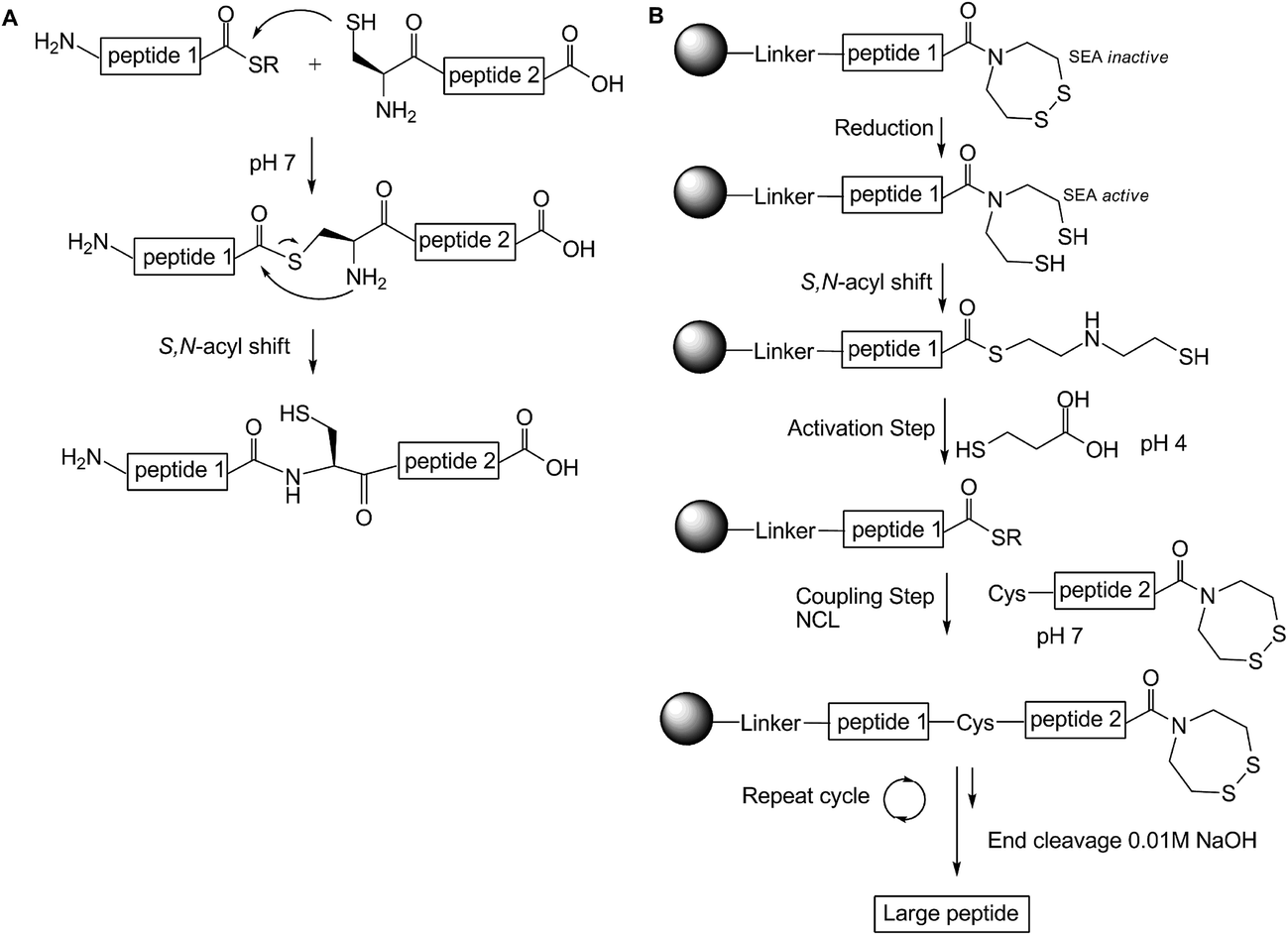
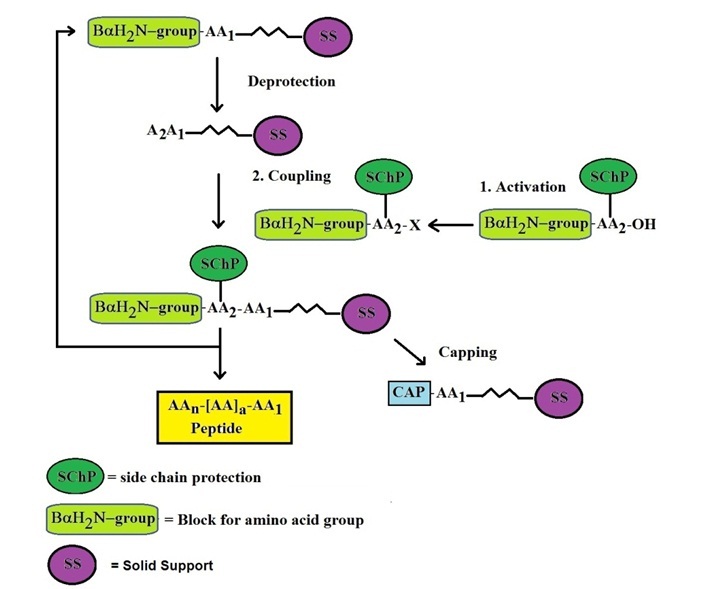
Solid-phase peptide synthesis: an overview focused on the preparation of biologically relevant peptides - RSC Advances (RSC Publishing) DOI:10.1039/C4RA02458C


![Schematic illustration of Solid Phase Peptide Synthesis [27] | Download Scientific Diagram Schematic illustration of Solid Phase Peptide Synthesis [27] | Download Scientific Diagram](https://www.researchgate.net/profile/Serap-Derman/publication/283498406/figure/fig1/AS:614207926063137@1523449989725/Schematic-illustration-of-Solid-Phase-Peptide-Synthesis-27_Q640.jpg)