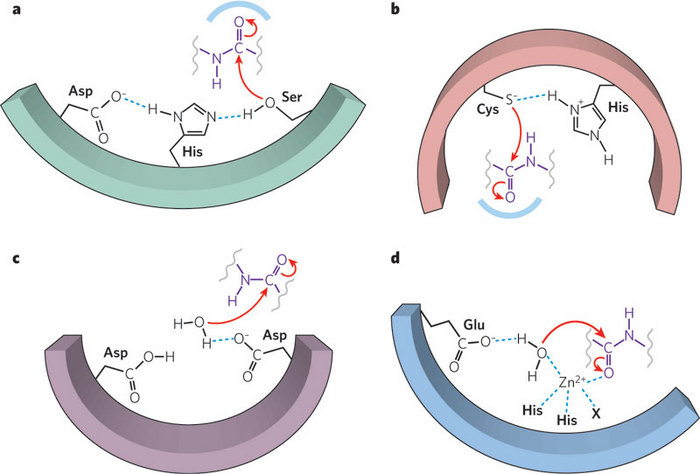
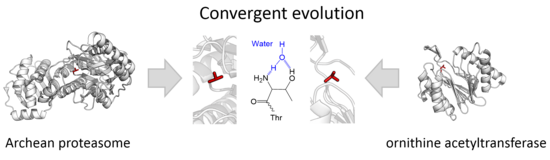
Why Does Threonine, and Not Serine, Function as the Active Site Nucleophile in Proteasomes?* - Journal of Biological Chemistry

Proteasome inhibitors mechanism; source for design of newer therapeutic agents | The Journal of Antibiotics

Cartoon showing substrate/inhibitor residues (P) and protease binding... | Download Scientific Diagram

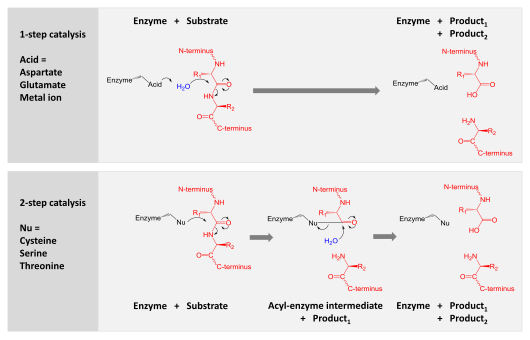
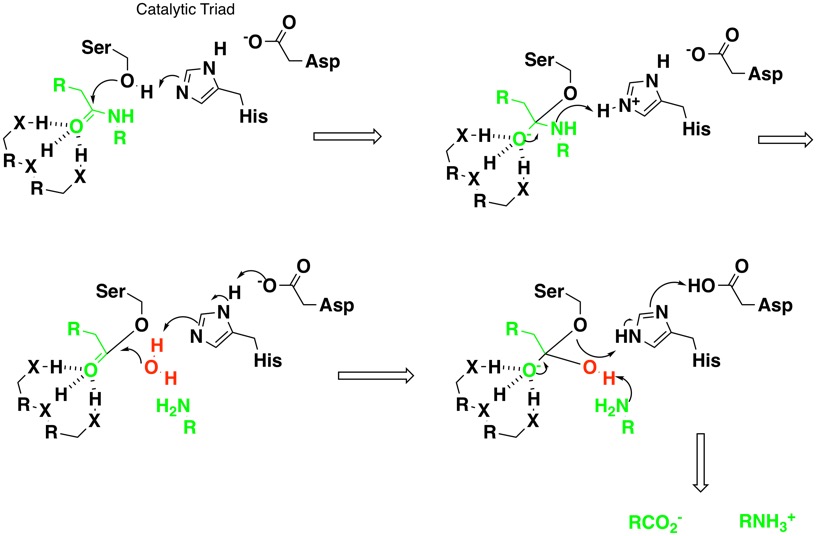
Intrinsic evolutionary constraints on protease structure, enzyme acylation, and the identity of the catalytic triad | PNAS

Cysteine proteases as therapeutic targets: does selectivity matter? A systematic review of calpain and cathepsin inhibitors - ScienceDirect
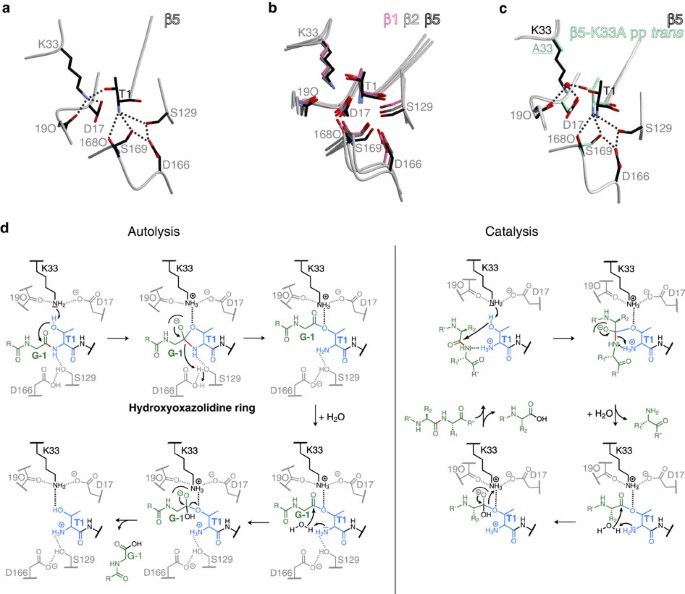
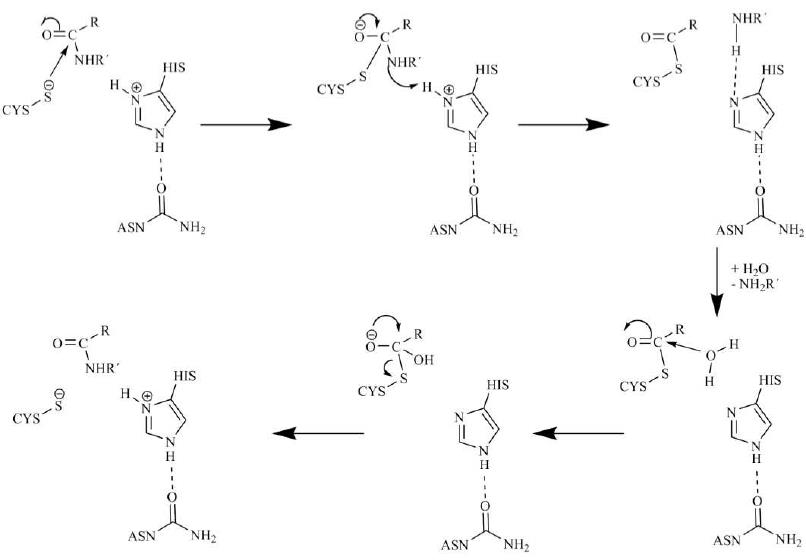
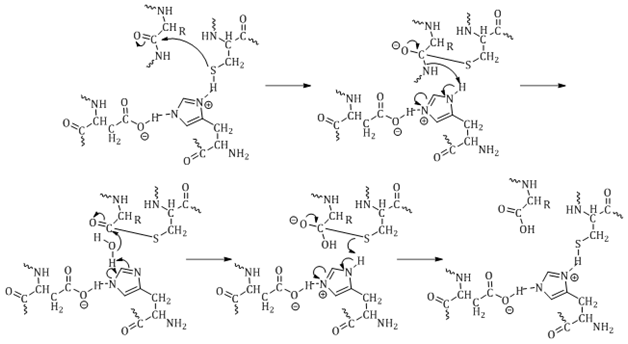
A unified mechanism for proteolysis and autocatalytic activation in the 20S proteasome | Nature Communications